Expanded Diagnoses Outside of Specialized Amyloidosis Centers: Results from the Amyloidosis Research Consortium’s 2022 Community Survey

Publications
A selection of key works.
The Amyloidosis Research Consortium (ARC) has an end-to-end research model focused on accelerating the development of and access to new, effective treatments. Our scientific publications have helped build a strong backbone for continued research in our mission to extend and improve the lives of those living with amyloidosis.
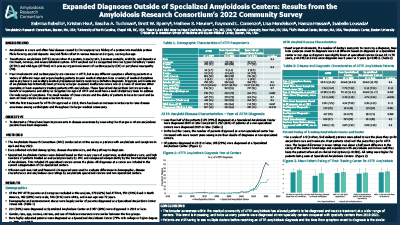
2022 Affordability Survey
Factors associated with financial toxicity in patients with ATTR: results from ARC’s ATTR Treatment Affordability Patient & Caregiver Survey in 2022.
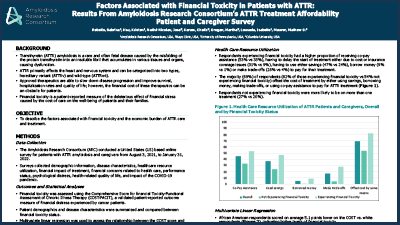
2021 ATTR-QOL Questionnaire Presentation
Development of a novel, ATTR-specific patient-reported outcome measure

2019 Recommendations to Improve Diagnosis
Expert consensus recommendations to improve diagnosis of ATTR amyloidosis with polyneuropathy

2019 ARC Research Roadmap
Identifying, improving, and accelerating a drug development pathway for the systemic amyloidosis diseases

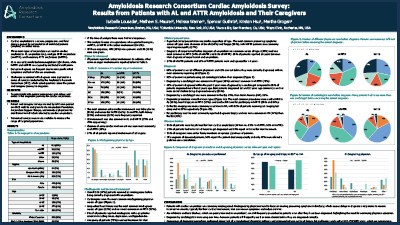
2019 Cardiac Amyloidosis Survey
Amyloidosis Research Consortium Cardiac Amyloidosis Survey: Results from Patients with AL and ATTR Amyloidosis and Their Caregivers
2016 Voice of the Patient Report
Summary report resulting from an EL-PFDD meeting, a parallel effort to the U.S. Food and Drug Administration’s (FDA’s) PFDD Initiative

2015 Patient Experience Survey
Light Chain Amyloidosis: Patient Experience Survey from the Amyloidosis Research Consortium




